The DRAGON project aims to use AI and machine learning to develop a decision support system capable of delivering more precise coronavirus diagnosis and predictions
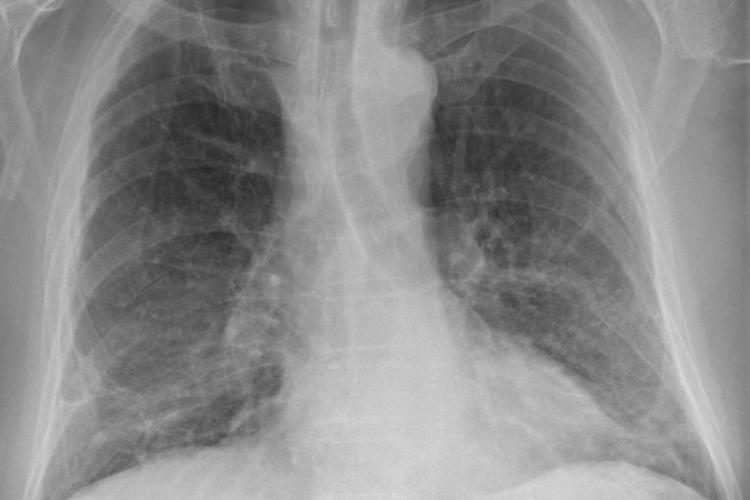
Imagine the scenario. It’s 2022, and although vaccines and treatments have dramatically reduced the number of people catching COVID-19, the disease continues to circulate in communities around the world. And you’ve got symptoms – you have a fever, and you’re so short of breath you decide to go to the hospital to get checked out. On arrival, they isolate you and run standard tests, including a chest x-ray and CT (computed tomography) scan.
What happens next? Well, the doctor plugs your test results, including the scans, into an AI-based decision support tool that compares your results with those of thousands of other people. After just a few minutes, the result pops up on the doctor’s screen. ‘Provisional diagnosis – Consistent with coronavirus pneumonia,’ the message reads. You’re quickly admitted to intensive care and given a treatment that has been shown to be effective for patients like you.
Meanwhile, another patient who came in with similar symptoms (aching joints and shortness of breath) underwent the same tests but got different results. ‘Pattern consistent with pulmonary edema in an individual with emphysema,’ the AI tool suggests. Although the patient had no history of heart disease, the doctor runs more tests which confirm that the patient has indeed had a heart attack.
If this sounds like science fiction, the DRAGON project hopes it will be a reality in the not too distant future. The project aims to deliver an electronic decision support tool that will speed up clinicians’ ability to diagnose COVID-19 and predict patients’ prognosis, i.e. how the disease is likely to progress in a given patient. They also want to deliver digital health tools that will empower citizens to participate in their own diagnosis, care and monitoring.
‘We will develop tools (AI algorithms, app, etc.) and test them in several prospective clinical trials,’ says project coordinator Philippe Lambin of Maastricht University, adding that the project will ensure the tools are compliant with relevant legislation on data protection and medical devices.
The project’s timelines are ambitious. ‘The first version of the electronic decision support system will be operational in January 2022, followed closely by the first version of the application,’ says Professor Lambin. ‘The final fully functional decision support system application will be available mid 2022 and will be continually updated after that.’
The project outputs will have to be approved by regulators before they can be used in healthcare, but Professor Lambin is confident that the consortium will overcome this challenge.
‘Many of the partners have established workflows and processes for regulatory approval and indeed the earliest outputs will move through these quickly,’ he explains. ‘One of the advantages of conducting this project in a highly interactive and cohesive consortium is that the partners can benefit from each other's experience and knowledge in moving through regulatory barriers.’
