How IMI works
This page explains how our top level objectives, as set out in the IMI1 and IMI2 legislation and the Strategic Research Agenda guide the choice of topics for Calls for proposals and, ultimately, the selection of projects.
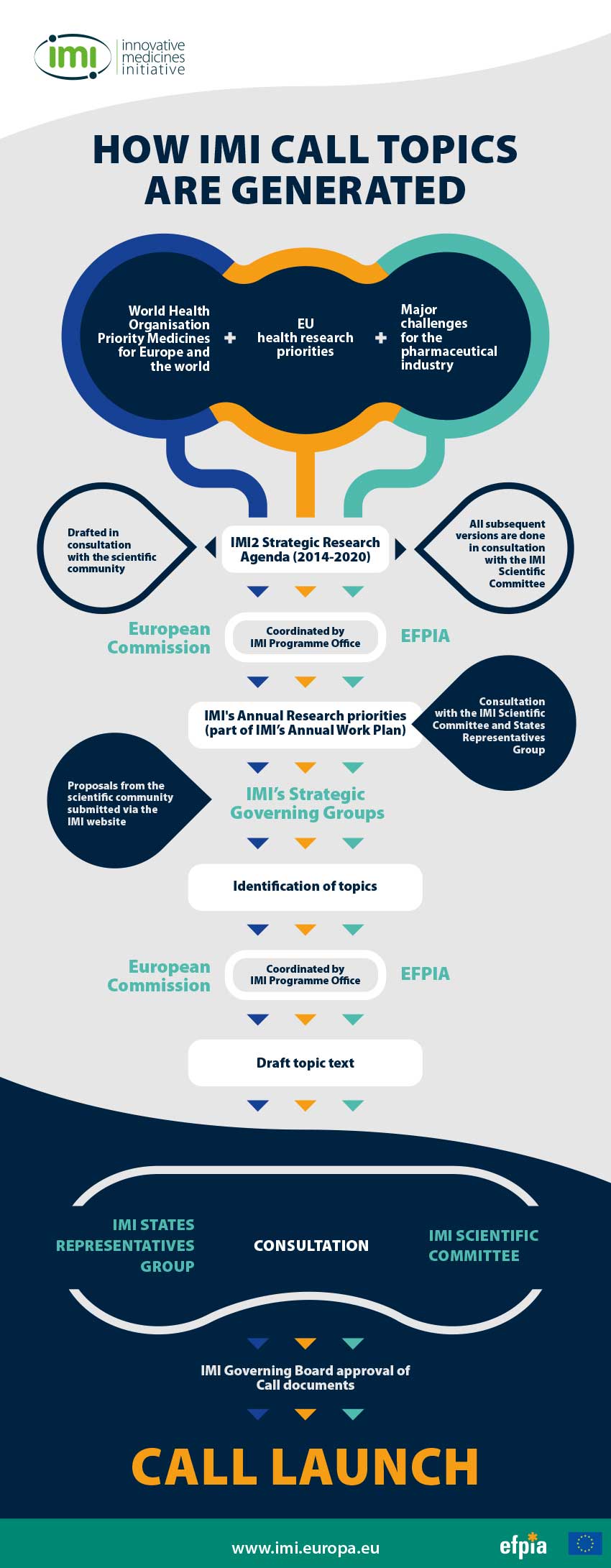
From top level objectives to annual priorities
Our top level objectives are set out in the legislation creating IMI1 and IMI2. IMI also has a multi-annual Strategic Research Agenda (SRA), which sets out our priority areas in more detail. Every year, IMI draws on its legislation and the SRA to set out annual research priorities. These form part of the Annual Work Plan, which is approved by the Governing Board and published online. These annual priorities are based on the need for collaboration in complex areas of biomedical research and innovation, and are a result of consultations between EFPIA companies, the European Commission and the other stakeholders.
From annual priorities to projects - how topics are generated
Drawing on the annual priorities, a consortium of EFPIA companies and, in some cases, other large companies or organisations active in health research, agree on the need to work together and with other stakeholders on a specific issue.
Who can submit an idea for an IMI Call topic?
Individual EFPIA member companies, an IMI Strategic Governing Group (SGG), an Associated Partner or third parties, may submit ideas for our topics. Third party ideas can be submitted via the specific form available on our website.
All third party ideas are shared with the relevant SGG or, if they are not in the SGGs scope, directly with the EFPIA research group. If a third party idea is aligned with the IMI Strategic Research Agenda and supported by EFPIA, then it may develop into a call topic led by an EFPIA/Associated Partner proponent. The third party submitters may not participate in development of a topic text if they wish to apply and receive funding when the corresponding call is issued.
From idea to topic text and budget
The EFPIA/Associated Partner proponent of the idea invites other companies or organisations to join and contribute to the preparation of a topic text. The topic text describes the challenge, scope, deliverables, the industry consortium and contributions, and the resources and skills expected from public sector applicants.
The drafting process allows companies to align and to define the pre-competitive space for collaboration. At this point companies also start identifying in-kind contributions (activities, resources and their indicative cost) that will be invested in the project. The topic writers are supported by the IMI scientific officers, who provide scientific input as well as guidance on our rules and processes.
In certain challenging areas where external expertise may be necessary, the IMI Programme Office may organise, usually at an early stage of the idea generation, a workshop to gain external input for the topic. New concepts are also sometimes subject to consultations online or during our annual IMI Stakeholder Forum.
Once a draft topic text is ready, it is submitted to the EFPIA research group that ensures that the proposed topic is in line with our objectives and adequately balances societal and industrial agendas. EFPIA will also review the topic robustness and resources (number of companies, in-kind commitments, etc.).
The consultation process and the Governing Board decision
Each topic text is subject to a formal consultation with the European Commission (EC), the IMI States Representatives Group (SRG) and the IMI Scientific Committee (SC).
All comments and questions are collated by the IMI Programme Office and shared with the topic writers. The drafting team prepares responses to each comment and the IMI office sends them back to the EC, SRG and the SC.
In most cases, questions and suggestions lead to subsequent adjustments to the topic text before a final legal check and proofreading by the IMI office and the launch of the IMI Governing Board approval process.
The final decision on whether a topic will be part of a call is the responsibility of the IMI Governing Board. Following the Governing Board’s green light, IMI launches a Call for proposals on its website and the European Commission’s Participant Portal.
Following an open, competitive evaluation process involving independent experts and Governing Board approval, the project is launched.
Linking projects with priorities
Looking over the list of IMI projects, it is clear that we have always maintained a strong focus on priority disease areas, where safe, effective treatments are lacking, and/or where the impact on public health is greatest. For example, we have a large portfolio of projects on infectious diseases, with dedicated programmes on antimicrobial resistance and Ebola and related diseases. Brain disorders, an area where drug development takes even longer and costs even more than for other areas, are also well represented in IMI, with projects on Alzheimer’s disease, schizophrenia, depression, chronic pain, and autism. Diabetes is another area where we are particularly active, and where we increasingly work in partnership with the patient organisation JDRF. In addition, we have a number of projects on cancer.
More broadly, we have many projects addressing cross-cutting issues in medicines research and development, and here results will help to improve the development of new treatments in all disease areas. For example, we have projects on drug and vaccine safety, big data and knowledge management, the sustainability of chemical drug production, the use of stem cells for drug discovery, drug behaviour in the body, and the creation of a European platform to discover novel medicines. In addition to research projects, we support education and training projects.